Noninvasive Therapy for Cartilage Regeneration
health medicine and biotechnology
Noninvasive Therapy for Cartilage Regeneration (MSC-TOPS-96)
Pulsed electromagnetic field device can restore life to damaged joints
Overview
Innovators at NASA Johnson Space Center researching time-variance magnetic field (TVMF) therapies have developed a pulsed electromagnetic field (PEMF) device that can alleviate cartilage degradation in synovial joints by promoting the growth of new cartilage.
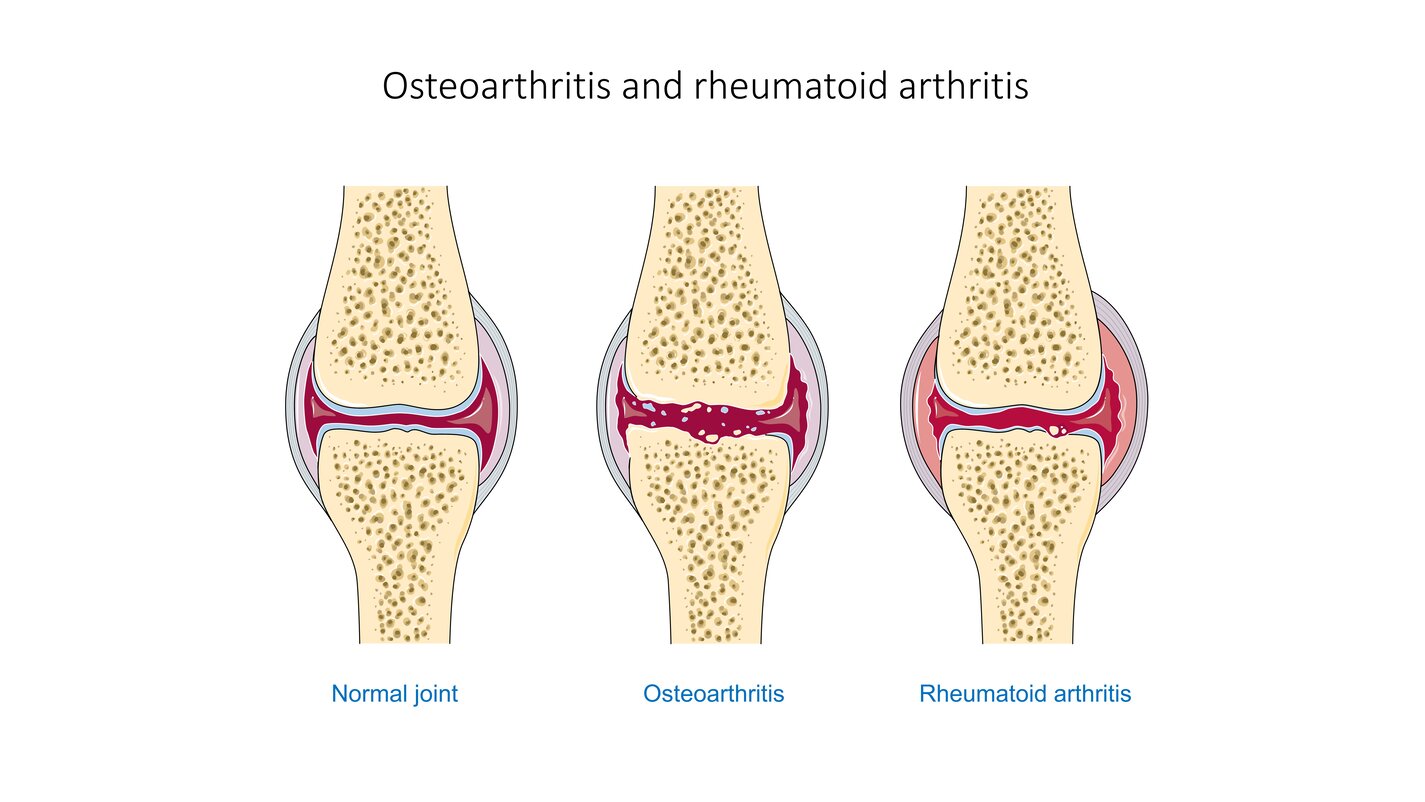
Joint disorders, whether induced by rheumatism, joint dysplasia, trauma, or surgery, often degrade cartilage and result in intense patient pain. Noninvasive and painless regeneration of a patients own tissue offers fewer side-effects than surgical joint replacement or tissue engineering procedures. The PEMF device could simply be wrapped around synovial joints where cartilage-degrading inflammation is located.
This pulsed electromagnetic field technology is at a technology readiness level (TRL) 6 (system/sub-system model or prototype demonstration in an operational environment) and the related patent is available for licensing. Please note that NASA does not manufacture products itself for commercial sale.
The Technology
Research has shown that exposure of mammalian cartilage and bone tissue to tuned magnetic fields modifies genetic regulation at a cellular level. PEMF therapy relies on modulation and resonance of weak metals (ions) such as Ca2+, K+, Li+, and Mg2+ which can be made to move at the sub-cellular level when exposed to magnetic flux. This NASA technology is a device and method for modifying genetic regulation of cartilage and bone in response to PEMF therapy and may serve as the basis for development of novel therapies for cartilage diseases.
In initial studies, cultured human chondrocyte cells (HCH) from patients with early-stage osteoarthritis were exposed to PEMF stimulation using a variety of tuned electro-magnetic pulse characteristics such as flux magnitude, slew rates, rise and fall times, frequency, wavelength, and duty cycle. Waveforms used in testing were monophasic, bi-phasic, square, sinusoidal, and triangular in nature. Frequencies were generally low, ranging from 6-500 Hz, and the waveforms used high rising and falling slew rates on the order of Tesla/sec, promoting pulses or bursts.
Cellular catabolic and anabolic gene expression analyses comprised of fold-change (in expression) were accomplished by a survey of 47,000 human genes using an AFFYMETRIX Gene Array. Results show that variation of waveform used in PEMF therapies, independent of flux intensity, influences genetic regulation of HCH from patients with early-stage osteoarthritis.

Benefits
- Non-invasive: PEMF therapy requires no surgery or invasive procedure
- Regenerative: PEMF therapy relies on upregulation of genetic factors to promote growth and restoration of compromised cartilage
- Potential for FDA approval: PEMF therapeutic devices have been approved by the FDA for use in the stimulation of bone regrowth in artificial joint disunions
Applications
- Medicine: treatment of cartilage degenerative joint disorders in patients resulting from rheumatism, trauma, or surgery
Technology Details
health medicine and biotechnology
MSC-TOPS-96
MSC-24541-1
MSC-24541-2
MSC-24541-3
|
Tags:
|
Similar Results

Ionic Magnetic Resonance Tailors Animal Cells/Tissues
The apparatus comprises a randomized gravity vector multiphasic culture system with a self-feeding growth module, an optionally disposable nutrient module, and a removable AIMR chamber that delivers a pulsating multivariant field to the contents of the culture system. It produces overlapping or fluctuating alternating ionic magnetic resonance frequencies at one or more modal intervals ranging from about 7.8 Hz to about 59.9 Hz to the cell chamber. The apparatus may yield better regulation that can be manipulated to allow for increased rate of cell growth, faster differentiation, increased cell fidelity, and the induction or suppression of selective physiological genes involved in directing cellular differentiation and dedifferentiation.
The use of an AIMR field may provide a significant improvement over existing bioreactors, including pulsating electromagnetic field (PEMF) and time-variance electromagnetic field (TVEMF) cellular growth induced systems, in that AIMR incorporates the modulation of cellular transcription. The AIMR system utilizes pre-sterilized disposable modules and a removable alternating ionic magnetic resonance chamber, reducing the hazard for contamination, allowing scientists to implement physiological and homeostatic parameters similar to a naturally occurring physiological system.

Electroactive Scaffold
Current scaffold designs and materials do not provide all of the appropriate cues necessary to mimic in-vivo conditions for tissue engineering and stem cell engineering applications. It has been hypothesized that many biomaterials, such as bone, muscle, brain and heart tissue exhibit piezoelectric and ferroelectric properties. Typical cell seeding environments incorporate biochemical cues and more recently mechanical stimuli, however, electrical cues have just recently been incorporated in standard in-vitro examinations. In order to develop their potential further, novel scaffolds are required to provide adequate cues in the in-vitro environment to direct stem cells to differentiate down controlled pathways or develop novel tissue constructs. This invention is for a scaffold that provides for such cues by mimicking the native biological environment, including biochemical, topographical, mechanical and electrical cues.

Hall Effect Thruster Technologies
Used for propelling Earth-orbiting satellites and deep-space robotic vehicles, the HET gets its name because it traps electrons with an intense radial magnetic field in an azimuthal Hall current moving around the circumference of an annular ceramic channel. The electrons in the circulating Hall current ionize the onboard propellant - the inert gas xenon - and create an ionized plasma. The xenon plasma is then accelerated axially, via an applied electric field along the coaxial channel, to an exit velocity of up to 65,000 miles per hour to produce thrust. The interaction of the accelerated plasma and the downstream edge of the channel, where the plasma is the most energetic, results in erosion of the surrounding magnetic system used to generate the plasma. One of NASA Glenn's novel designs relies on an azimuthally symmetric configuration that minimizes radial magnetic fields at the discharge chamber walls. This configuration completely shields the walls of the discharge chamber from the high-energy plasma ions. With regard to the discharge-channel-wall replacement innovation, an actuator can be configured to extend the discharge chamber along the centerline axis. The actuator can be either mechanical or programmable. In either case, the sleeve can be extended while an upstream portion of the discharge chamber remains stationary, thereby preventing plasma exposure. These novel designs increase the efficiency and extend the lifetime of the HET to five times that of unshielded thrusters, enabling a new era of space missions.

Electric Field Imaging System
The EFI imaging platform consists of a sensor array, processing equipment, and an output device. By registering voltage differences at multiple points within the sensor array, the EFI system can calculate the electrical potential at points removed from the sensor. Using techniques similar to computed tomography, the electrical potential data can be assembled into a three-dimension map of the magnitude and direction of electric fields. Since objects interact with electric fields differently based on their shape and dielectric properties, this electric field data can then be used to understand shape,
internal structure, and dielectric properties (e.g., impedance, resistance) of objects in three dimensions.
The EFI sensor can be used on its own to see electric fields or image electric fieldemitting objects near the sensor (e.g., to evaluate leakage from poorly shielded wires or casings). For evaluation of objects that do not produce an electric field, NASA has developed generator that emits a low-current, human-safe electrostatic field for snapshot evaluation of objects. Additionally, an alternative EFI system optimized to evaluate electric fields at significant distances (greater than 1 mile) is being developed for weather-related applications.

Wireless Electrical Devices Using Floating Electrodes
The technology presents a fundamental change in the way electrical devices are designed, using an open circuit in conjunction with a floating electrode, or an electrically conductive object not connected to anything by wires, and powered through a wireless device. This system uses inductor-capacitor thin-film open circuit technology. It consists of a uniquely designed, electrically conductive geometric pattern that stores energy in both electric and magnetic fields, along with a floating electrode in proximity to the open circuit. When wirelessly pulsed from the handheld data acquisition system (U.S. Patent Number 7,159,774, Magnetic Field Response Measurement Acquisition System), the system becomes electrically active and develops a capacitance between the two circuit surfaces. The result is a device that acts as a parallel plate capacitor without electrical connections.



